AEMPS authorizes the initiation of the clinical trial of AP-2, Molefy Pharma’s therapeutic candidate for ALS
AEMPS authorizes the initiation of the clinical trial of AP-2, Molefy Pharma’s therapeutic candidate for ALS • The AEMPS authorizes the initiation of the Phase I clinical trial of AP-2, which will begin in April at Hospital Universitario de La Princesa with 70 healthy volunteers • AP-2, developed by Molefy Pharma based on CSIC research, targets the TDP-43 […]
Clinical Trials in Spain: A Strategic Opportunity to Accelerate Innovation in ALS

The development of new therapies for amyotrophic lateral sclerosis (ALS) does not rely solely on advances in biomedical research at the laboratory level. It also requires the ability to translate that knowledge into the clinical setting in an efficient, safe, and coordinated way. In this context, clinical trials in Spain have become a fundamental pillar […]
What does obtaining orphan drug designation in Europe mean and why is it key to innovation in ALS?

Orphan drug designation in Europe is a regulatory recognition granted by the EMA to treatments intended for rare diseases, such as ALS. This status provides key incentives—such as market exclusivity and scientific support—that reduce investment risk and accelerate therapeutic innovation, driving the development of new options for patients with urgent unmet medical needs.
The future of ALS research: why the coming years will be decisive

Amyotrophic lateral sclerosis (ALS) is one of the most complex and devastating neurodegenerative diseases of our time. For decades, biomedical research has progressed slowly, with limited therapeutic options and modest clinical impact. However, the landscape is changing. The coming years are shaping up to be a critical turning point for pharmaceutical research in ALS, driven […]
A Canary Islands Company Leads the Clinical Trial of a New Drug to Treat ALS

Following the discovery of several molecules by a CSIC research group, Molefy Pharma will become one of the first organizations worldwide to evaluate the viability of this new therapeutic approach. By Verónica Pavés. Santa Cruz de Tenerife. Originally published by El Día. A company based in the Canary Islands will lead the clinical trial of a promising new treatment aimed […]
Molefy Pharma receives Orphan Drug Designation (ODD) from the European Union for its AP-2 candidate in the treatment of amyotrophic lateral sclerosis (ALS)

The European Medicines Agency (EMA) has granted Molefy Pharma the Orphan Drug Designation (ODD) for its candidate AP-2, an innovative therapy developed for the treatment of amyotrophic lateral sclerosis (ALS). This designation is awarded to medicines with the potential to treat rare and severe diseases in the European Union, when they offer a significant benefit […]
ALS Association awards $1 million to Molefy Pharma to advance its clinical trial against ALS

Spanish biotech company receives backing from one of the leading global organizations in the fight against amyotrophic lateral sclerosis (ALS) ARQUIMEA, Spanish technology group and co-founder of the spin-off Molefy Pharma together with researchers from the CSIC, celebrates the award of a $1 million grant from the ALS Association (ALS.org) to accelerate the development of […]
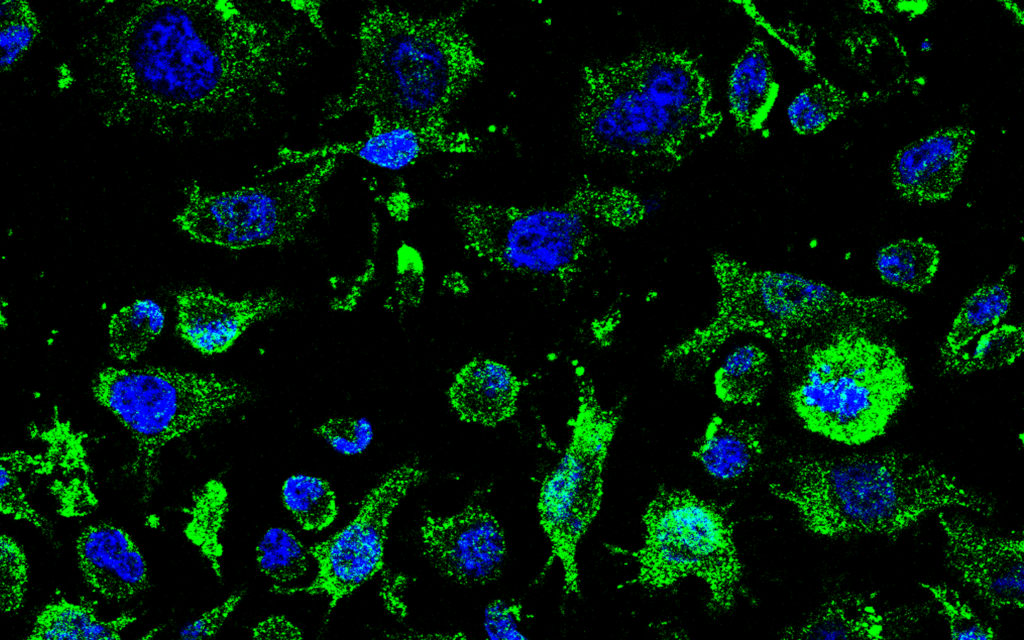
Avoid cell-to-cell disease transmission in ALS patient-based cellular model
Efficacy in transgenic TDP-43 mouse model and in ALS patient-based cellular model
Synthesis and first biological profile of the inhibitors