The Spanish Agency of Medicines and Medical Devices (AEMPS) authorizes the start of the clinical trial of AP-2, Molefy Pharma’s therapeutic candidate for ALS
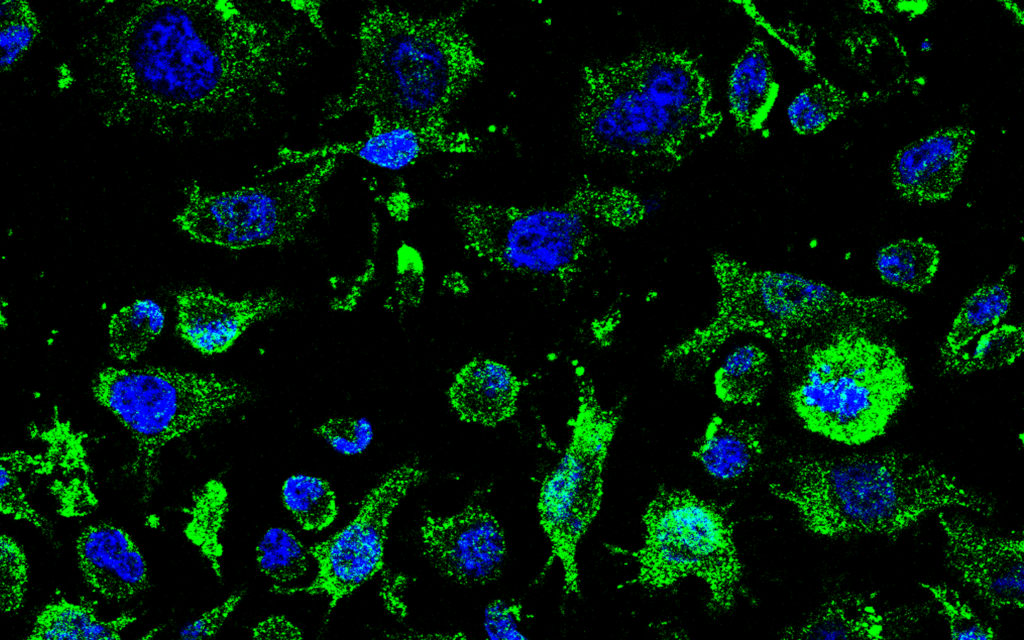
• The AEMPS authorizes the initiation of the Phase I clinical trial of AP-2, which will begin in April at Hospital Universitario de La Princesa with 70 healthy volunteers • AP-2, developed by Molefy Pharma based on CSIC research, targets the TDP-43 protein and has been granted orphan drug designation by the European Medicines Agency (EMA) Molefy Pharma, a […]
A Canary Islands Company Leads the Clinical Trial of a New Drug to Treat ALS

Following the discovery of several molecules by a CSIC research group, Molefy Pharma will become one of the first organizations worldwide to evaluate the viability of this new therapeutic approach. By Verónica Pavés. Santa Cruz de Tenerife. Originally published by El Día. A company based in the Canary Islands will lead the clinical trial of a promising new treatment aimed […]
Molefy Pharma receives Orphan Drug Designation (ODD) from the European Union for its AP-2 candidate in the treatment of amyotrophic lateral sclerosis (ALS)

The European Medicines Agency (EMA) has granted Molefy Pharma the Orphan Drug Designation (ODD) for its candidate AP-2, an innovative therapy developed for the treatment of amyotrophic lateral sclerosis (ALS). This designation is awarded to medicines with the potential to treat rare and severe diseases in the European Union, when they offer a significant benefit […]
ALS Association awards $1 million to Molefy Pharma to advance its clinical trial against ALS

Spanish biotech company receives backing from one of the leading global organizations in the fight against amyotrophic lateral sclerosis (ALS) ARQUIMEA, Spanish technology group and co-founder of the spin-off Molefy Pharma together with researchers from the CSIC, celebrates the award of a $1 million grant from the ALS Association (ALS.org) to accelerate the development of […]
Molefy Pharma launches new website to share breakthrough progress in ALS research and drug development
Santa Cruz de Tenerife, 9 de junio de 2025. Molefy Pharma, a biotech company born from the collaboration between ARQUIMEA and CSIC, has announced the launch of its new corporate website www.molefypharma.com , a platform designed to offer comprehensive insight into the company’s mission, scientific research, and advancements in the development of innovative treatments for […]
Molefy wins the eighth edition of the Investment Forum of the Science and Technology Park of Tenerife

Moley Pharma, a spin-off of the CSIC in collaboration 1with ARQUIMEA dedicated to finding a cure for amyotrophic lateral sclerosis (ALS) through the AP-2 molecule, has won first prize in the eighth edition of the Investment Forum of the Science and Technology Park of Tenerife. The event, held as part of the second edition of Hub Intech, […]
Molefy Pharma Showcased New ALS Research at ENCALS 2025 in Turin

Santa Cruz de Tenerife, 9 de junio de 2025. Molefy Pharma, a biotech company focused on developing innovative therapies for ALS and other neurodegenerative diseases, successfully presented three scientific papers at the ENCALS 2025 Meeting, held in Turin, Italy, reaffirming its commitment to advancing therapeutic innovation in Amyotrophic Lateral Sclerosis (ALS). The European Network to […]