AEMPS authorizes the initiation of the clinical trial of AP-2, Molefy Pharma’s therapeutic candidate for ALS
AEMPS authorizes the initiation of the clinical trial of AP-2, Molefy Pharma’s therapeutic candidate for ALS • The AEMPS authorizes the initiation of the Phase I clinical trial of AP-2, which will begin in April at Hospital Universitario de La Princesa with 70 healthy volunteers • AP-2, developed by Molefy Pharma based on CSIC research, targets the TDP-43 […]
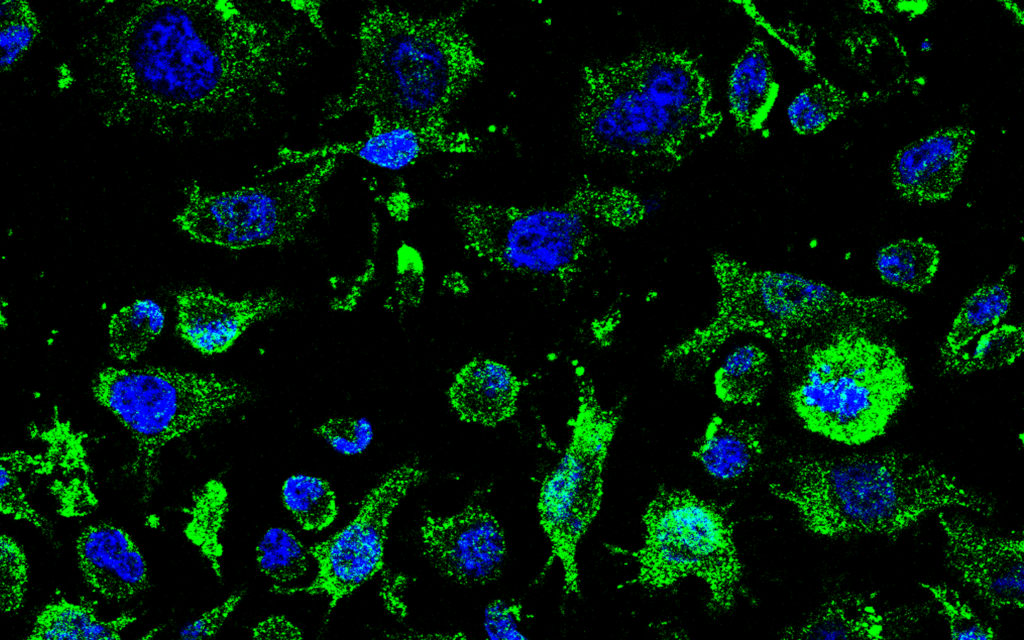
Molefy Pharma Showcased New ALS Research at ENCALS 2025 in Turin

Santa Cruz de Tenerife, 9 de junio de 2025. Molefy Pharma, a biotech company focused on developing innovative therapies for ALS and other neurodegenerative diseases, successfully presented three scientific papers at the ENCALS 2025 Meeting, held in Turin, Italy, reaffirming its commitment to advancing therapeutic innovation in Amyotrophic Lateral Sclerosis (ALS). The European Network to […]